
Petrochemical Industry Solutions
SIFT-MS presents a breakthrough in the detection, quantitation and tracing of petroleum hydrocarbons. This brochure outlines several SIFT-MS-based petrochemical industry solutions provided by Syft Technologies including mud logging, sour gas detection, oil and gas prospecting, leak detection, and fence line monitoring.

Hydrogen Sulfide in Natural Gas
We present the results of the first study where SIFT-MS was used to detect and quantify hydrogen sulfide in natural gas and liquefied petroleum gas (LPG). SIFT-MS very effectively detects and quantifies hydrogen sulfide to mid part-per-billion levels in natural gas and LPG (due to the need for dilution). The degree of souring of natural gas, in addition to determining its light to medium hydrocarbon composition, can be determined.
Instant Detection of Non-Conforming Raw Materials Using SIFT-MS: A Semiconductor Industry
SIFT-MS can continuously analyze volatiles that indicate non-conformance of raw materials, such as bulk gases utilized in semiconductor manufacture. This webinar describes how SIFT-MS can be deployed in both targeted and un-targeted operational modes to enhance quality assurance and increase yields, via continuous monitoring.

Semiconductor Bulk Gas Analysis
Real-time monitoring of gas streams using selected ion flow tube mass spectrometry (SIFT-MS) provides continuous product quality feedback. This application note describes how SIFT-MS SCAN analysis coupled with multivariate statistical analysis readily detects untargeted defects in semiconductor manufacturing “bulk gas.”

Cleanroom Air Quality Monitoring
SIFT-MS is a unique analytical solution that provides comprehensive, high-sensitivity detection of volatile organic and inorganic gases (including HCl, HF, and SOx) within seconds. As a single, comprehensive instrument with rapid analysis, SIFT-MS provides great economic benefit. This webinar describes the application of SIFT-MS to AMC monitoring – from the front-opening unified pod (FOUP) to the clean room.
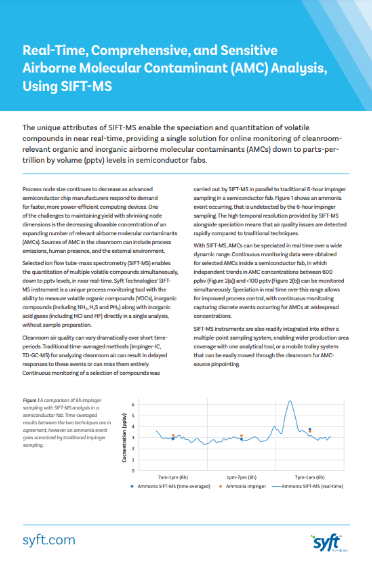
Semiconductor AMC Monitoring
The unique attributes of SIFT-MS enable the speciation and quantitation of volatile compounds in real-time, providing a single solution for online monitoring of clean room relevant organic and inorganic airborne molecular contaminants (AMCs) down to parts-per-trillion by volume (pptv) levels in semiconductor fabs. Learn how to detect AMCs when they happen and never miss a contamination event again.


An Evaluation of SIFT-MS for Instrument-Based Sensory Analysis: A Packaging Case Study
This webinar addresses chemical and sensory-like analysis using SIFT-MS in the context of odors from paperboard packaging. SIFT-MS is a powerful tool for rapid instrument-based sensory screening.


Analysis of Volatile Emissions from Polymers and Paperboard
This webinar describes a recent addition to the portfolio of techniques available for routine sample analysis: SIFT-MS. SIFT-MS enables rapid, quantitative analysis of VOCs across the full spectrum of packaging materials. Detailed case studies will describe applications in polymeric and paperboard emissions analysis.

Routine Analysis 3: SIFT-MS Method Development and Validation
In this webinar, we discuss how analytical method validation is approached for SIFT-MS so that data collected can be used to support regulatory requirements. This is achieved by applying a strategic approach in accordance with ICH Q2(R1) guidelines. Finally, we will illustrate SIFT-MS method validation using two case studies: 1) common toxic volatiles in water, 2) BTEX in soil following methanolic extraction.

Residual Monomer Analysis
In this paper, the unique capabilities of SIFT-MS are illustrated for residual monomer analysis, using formaldehyde emissions from polyoxymethylene (POM) polymer to illustrate. SIFT-MS provides significant throughput increases for both testing laboratories and quality assurance/quality control (QA/QC) in a process environment.
